When you need more than just code
At ImFusion, we don’t just build software — we bring proven domain expertise and hands-on support. From clinical prototypes to regulatory submissions to co-authored publications, we help bridge the gap between concept and deployment.
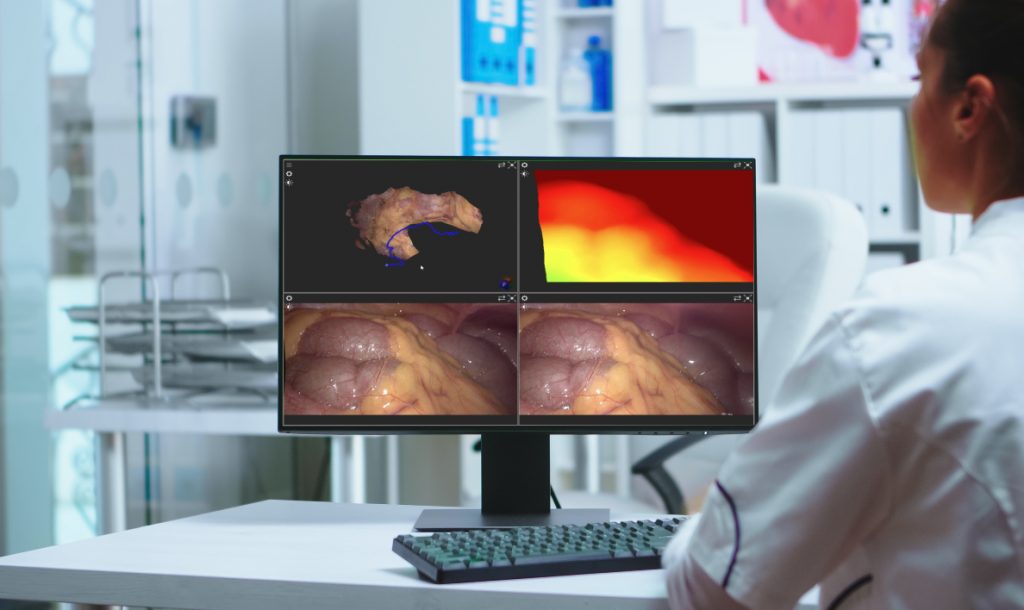

Consulting
We work closely with healthcare innovators to design and deliver custom imaging software. From prototyping clinical applications to optimizing algorithm performance and integrating with hospital infrastructure, our engineers act as an extension of your team — accelerating development without compromising reliability.
No task is too complex, and confidentiality is always guaranteed. We’d love to hear about your project and show you how we can help you get there — faster.
Regulatory Compliance
Bringing medical software to market means meeting strict regulatory standards. We provide expert support throughout the CE and FDA submission process, including documentation aligned with IEC 62304, risk and cybersecurity analysis, and structured reporting for AI/ML models.
The FAQ section below covers the main questions our partners had before working with us.


See our white papers

Ensuring Quality - The Key Elements of Technical Documentation for Medical Devices
An overview of our documentation process, ensuring IEC 62304 compliance.

Getting the Green Light - Regulatory Framework for Software as a Medical Device
A guide to navigating EU and FDA regulations for software, with focus on cybersecurity and evolving standards.

Academic Support
We collaborate with research groups worldwide to accelerate imaging innovation. Whether it’s building custom prototypes, preparing demos for grant applications, or co-authoring scientific papers, we support your team with high-performance tools (at a friendly price) and practical guidance — from project kickoff through publication.
We regularly celebrate our latest publications and collaborators on our LinkedIn company page so follow us! Additionally, we keep an extensive list of all the publications in which we were so lucky to be involved.
FAQ
1. Can ImFusion provide technical documentation for regulatory submissions?
2. Are your development processes compliant with ISO 13485?
Absolutely. Our quality management system has been certified to EN ISO 13485:2016 since 2016 and undergoes annual audits. All software development activities follow clearly defined SOPs, including risk management, configuration control, and document versioning.
3. Can you assist with cybersecurity?
We conduct cybersecurity risk analyses for software developed by using the STRIDE model and prepare documentation that meets IEC 81001-5-1, MDR Annex I, and FDA cybersecurity guidance. This includes risk mitigation strategies, data flow analysis, and support for secure software design.
4. What documentation do you provide for your AI-powered medical software?
For AI-powered medical software, we supply model reports detailing architecture, training datasets, performance metrics (e.g. accuracy, F1 score), and limitations. These documents align with EU AI Act guidance and FDA recommendations for SaMD.
5. What is ImFusion’s role in the post-market phase?
While the manufacturer retains full regulatory responsibility, we support post-market surveillance activities such as bug tracking, risk re-evaluation, and maintenance documentation updates. We also update traceability links if software is modified post-deployment.
No one likes to work alone
Whether you’re developing software for a commercial device or an academic study, we can support you across development, documentation, and regulatory processes.
